
Methanol (methyl alcohol wood alcohol) is used extensively as a solvent for lacquers, paints, varnishes, cements, inks, dyes, plastics, and various industrial coatings. The air odor threshold has been reported as 1500 ppm (approximately 2000mg/m3), much higher than the occupational guidelines. Methanol is a clear, colorless liquid with a characteristic pungent odor (NTP NIEHS web accessed ). Methanol is easily converted into important organic synthesis intermediates such as methyl carboxylate, methyl chloride and methylamine, and it is also applied as important organic solvents, extraction agents and alcohol denaturants. Therefore, it is largely used in the synthesis of formaldehyde. The three hydrogen atoms on a carbon atom with a hydroxyl group can be oxidized, orderly generating formaldehyde, formic acid, and carbon dioxide. Methanol has the general properties of a primary aliphatic alcohol. Once swallowed, it can make the eyes blind and even cause death. Its severe toxicity can damage the optic nerve. 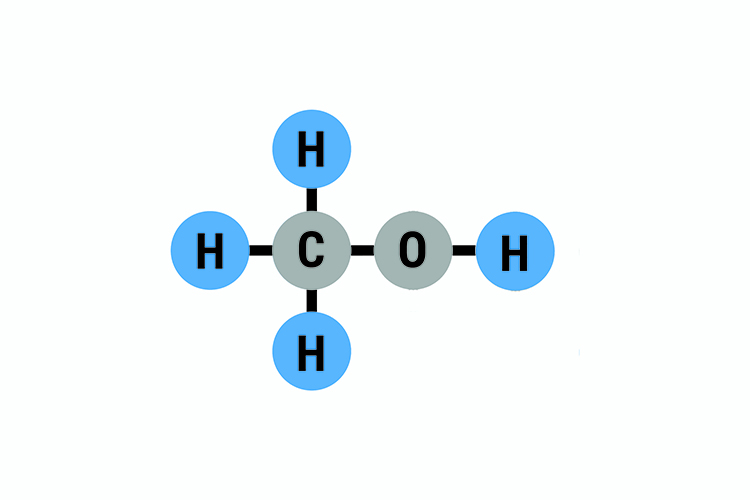
Soluble in water and most organic solvents. It is a colorless, flammable, irritating liquid with a boiling point of 64.7☌, a melting point of -93.90☌, and a relative density of 0.7913. Faint alcohol odor detectable at 4 to 6000 ppm (mean = 160 ppm)Ĥ.99 at 25 ☌ (headspace-GC, Gupta et al., 2000)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
